Plastic parts such as medical devices and equipment need to be manufactured in cleanrooms as they need to be sterile. That means protection from dust, chemical vapors, aerosol particles, airborne microbes, and other contaminants.
Maintaining cleanrooms is important for product quality, integrity, and safety cannot be compromised under any circumstances. In this article, we explain in detail how to maintain cleanrooms for manufacturing.
What is Cleanroom Injection Molding?
In order to maintain cleanroom standards, it is essential to follow cleanroom protocols! Responsible injection molders that manufacture medical devices, ensure that they follow and adhere to the stringent protocols of cleanroom standards. Injection molders must ensure that the various variables are maintained in order to meet compliance.

It is imperative that Clean rooms control various types of environmental conditions such as humidity, temperature, and pressure to ventilation. Maintaining a cleanroom for injection molding and manufacturing medical devices is optimized by ensuring the following:
- Clean room surfaces should be always impervious.
- Surfaces and equipment need to be cleaned regularly to avoid any contamination or dust to settle
- Strict control upon access to the clean room and apparel needs to be ensured.
- Higher pressure than neighboring environments (so that nothing gets sucked in)
We have discussed these protocols and various environmental personnel guidelines in detail in this section:
Cleanroom Maintenance Protocols
The first parameter that must be paid attention to is the quality control systems. Systems such as HEPA filters and machine hoods. These quality control systems monitor, manage and remove particulates and dust particles. If not controlled, it can cause particles to enter the environment and result in failing the ISO clean room standards.

- The next parameter that needs to be paid strict attention to is procedure documentation. Procedure documentation physically traces the process lifecycle, mold pull cycles, equipment testing, and product testing. This is essential, especially in the event of repeated cycles, routine checks, customer requests, or procedural deviation.
- Another parameter that needs to be ensured is the work environment controls. This helps to test air quality for purity and conduct routine equipment checks to ensure that there are no contaminants.
- The next protocol that injection molders should adhere to is the risk management protocol. Risk management can be implemented through specific analysis modules such as FTA, abbreviated for fault tree analysis. FEMA, abbreviated for failure mode and effect analysis, and HACCP, abbreviated for hazard analysis and critical control point.
These protocols help injection molders identify acceptable microbial limits and develop consistent contamination control assessments during manufacturing. - In addition, machine operators must ensure that every procedure is adhered to within a clean room. They must wear all types of PPE such as gloves, gowns, masks, and booties to avoid cross contamination.
Environment Personnel Guidelines
People can naturally introduce contaminants such as skin sheds, oils, sweat, and hair into the cleanroom environment thus, it is imperative that precautions are taken to avoid these contaminants from entering the cleanroom. In addition, human movement and behavior also need to be controlled in order to maintain the cleanliness of the cleanroom. We have listed some personnel guidelines that are necessary to keep a controlled environment so that the cleanroom environment is not put at risk:
- Personal items should be left outside the controlled environment. These include but are not limited to keys, wallets, phones, etc. If they need to be brought into the cleanroom for any reason, they should stay concealed beneath appropriate cleanroom garments.

- Eating, smoking, chewing gum in the cleanroom, or wearing makeup, perfume, etc. should be prohibited inside the cleanroom. These activities can unnecessarily introduce contaminants into the cleanroom thereby compromising its integrity.
- Running or moving more quickly than necessary shouldn’t be allowed in a cleanroom environment as fast motions can stir up particulates and lead to an increased risk of contamination.
- It should be ensured that if any personnel is unwell, then they should not enter the cleanroom as that can compromise the cleanroom environment.
- You must adhere to proper gowning and ungowning procedures. You should not let anyone enter the cleanroom if they are not appropriately covered. This entails clean and gloved hands, face masks, covered shoes, and safety glasses. In addition, any type of hair and facial hair need to be covered as stray hairs or hair oils can contaminate the products.
- Leaning on equipment or work surfaces should be avoided. When you or your garments touch one surface and then another, you transfer any contaminants between the two thus, you must limit the surfaces you touch to only those that are necessary.
- You should not touch your face or fidget with your hair with gloved hands. If you need to redo anything, you must exit the cleanroom and replace your gloves before re-entering the cleanroom.
How to Maintain Cleanroom Cleanliness?

Maintain the Right Temperature and Humidity
Most cleanroom environments require a relative humidity level to be maintained between 30% – 40% RH with an air temperature of 21°C / 69.8°F. This is generally an ideal environment to avoid many negative repercussions such as bacterial/fungal growth, corrosion, and static electricity. It is also ideal for employee comfort because if they tend to sweat or shiver, they may release more particles into the environment.
Maintain the Airflow
It is imperative to regulate the airflow in order to maintain a clean room. There are 2 types of air flows that are regulated to avoid the risk of contamination.
Positive Pressure Airflow
Positive pressure air flow is created when clean, filtered air is pumped through the ceiling into the cleanroom and is generally used to keep contaminants out of the cleanroom. If there is a leak, positive pressure airflow forces clean air into the cleanroom.
Negative Pressure Airflow
On the other hand, in a negative pressure cleanroom, all the outlets i.e. the windows and the doors are completely sealed. Thus, a lower pressure is created within the room. This results in outside air flowing into the cleanroom. Negative pressure is applied when you want to keep any possible contamination escaping from the cleanroom.
Cleanroom-Compliant Office Supplies
One of the most overlooked sources of contamination is common office supplies, such as mouse pads, notebooks, sticky notes, paper, and ID badge holders. Moving a pen from an office environment to a cleanroom can cause contamination thus, it’s important that these materials remain in your cleanroom. It’s also best not to post anything on the cleanroom walls, such as sticky notes as it can shed particles.
Consider Supply Packaging
The supply packaging that is used may not always be compliant. For instance: If you order nitrile cleaning gloves that are cleanroom-compliant, the cardboard box that dispenses them probably isn’t, as it could release particulate into the air thus, these errors need to be paid attention to.
Have the Right Cleaning Materials
It is important to ensure that the right cleaning materials are used to avoid the risk of contamination. Simply using a paper towel or cloth can compromise a cleanroom thus, wipes, swabs, and other cleaning supplies should be carefully evaluated for use in cleanrooms and as per the specific ISO Class. It’s also necessary to keep exclusive cleaning equipment such as brooms and mops. These supplies should be exclusive to your cleanroom and not used elsewhere in the facility.
Avoid Outside Personal Products
While procedures often cover standards for personal hygiene, it’s important that the cleanroom workers avoid make-up, perfume, and jewelry which are potential contaminants. Items such as food, beverages, candy, or gum should not be brought into the cleanroom.
Cleanroom Standards
Cleanroom standards are classified through ISO 14644-1 and it is defined by the degree of purity of the air which is based upon particle concentration per m³. This system classifies the cleanrooms through ISO 1-9 in which the highest purity is that of ISO class 1, whereas the lowest is ISO class 9. The following table depicts the Cleanroom Standards:
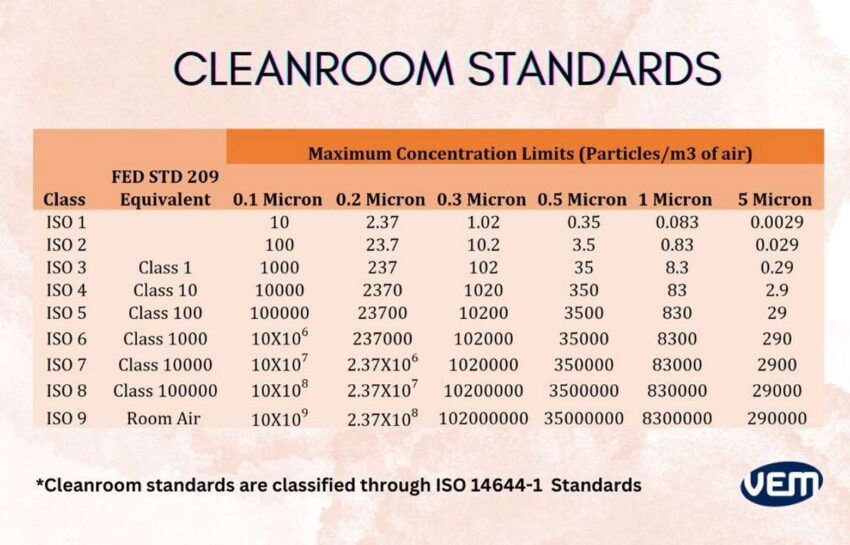
If you would like to learn more about cleanrooms, you can view this article where we explain in detail the requirements of cleanrooms.
Contact Us for a Cleanroom Injection Molding Partner
Our plastic injection molding and tooling facility in Thailand has various cleanrooms. Our team in Thailand has the expertise and is dedicated to providing you with a great manufacturing experience and exemplary service. Contact us and a team member will connect with you to resolve your queries.




